Main content
Top content
Confined and jacketed Macromolecules
1. Nanostructurized semi-interpenetrating networks
In cooperation with Prof. R. Kimmich, Sect. Nuclear Resonance Spectroscopy / University of Ulm
Complementary to polymer blending, the formation of interpenetrating networks (IPN) represents an approach towards the physical combination of immiscible polymers into one mesoscopically homogenous material. Due to the incompatibility of most polymers, IPN´s usually exhibit a microphase domain morphology, that is formed during IPN synthesis. Semi-interpenetrating networks of poly(ethylene oxide) (PEO) and highly crosslinked poly(methacrylate)s have been generated from solutions of PEO in mixtures of methacrylate monomers and dimethacrylate crosslinkers. Deep quenching of the solutions into the instable region results in microphase separation via a spinodal decomposition mechanism and subsequent crystallization of the PEO (Fig. 1.1a). This microstructure was permanently conserved by UV initiated polymerization of the monomers well below the melting temperature of the PEO. Peculiar morphologies consisting of percolating networks of cylindrical poly(ethylene oxide) aggregates with diameters of (10 +- 2) nm have been observed, independent of the molecular weight of the used poly(ethyleneoxide) (Fig. 1.2).
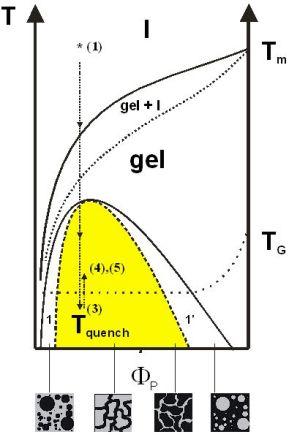
Figure 1.1a: Schematic depiction of a polymer / solvent phase diagram illustrating the preparation process of the semi-IPNs.
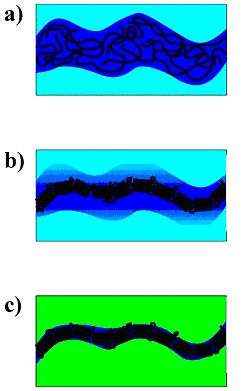
Figure 1.1b: Proposed mechanism for the formation of ‘thin’ PEO-tubes in crosslinked poly(methacrylate) matrices. a) Threat-like string of the PEO-rich phase at the begin of the spinodal demixing, b) crystallisation of the PEO in the threat, completely expelling the solvent, c) after polymerization of the solvent: thin embedded PEO tube.

Figure 1.2a: Overview - Polymerized PEO -10.000 (20wt%) / HEMA / EGDMA gel TEM micrograph, freeze-fracture / replica technique

Figure 1.2b: Detail - Polymerized PEO -10.000 (20wt%) / HEMA / EGDMA gel TEM micrograph, freeze-fracture / replica technique
2. Polymer / Amphiphile complexes
“Jacketed Macromolecules” - Complexation of Polybases with Wedge-Shaped Sulfonic Acids for the Synthesis of Supramolecular Cylinders
In cooperation with Prof. M. Möller and Dr. X. Zhu , DWI an der RWTH-Aachen , Pauwelsstr. 8, D-52056 Aachen, Germany
Supramolecular self-organization is a powerful tool to produce functional materials.[1] Recent effort in this field focused on the development of concepts to generate materials with well defined structures at the molecular level. One concept is based on complexation of polymers with low molecular weight amphiphiles by means of ionic interactions, coordinative- or hydrogen bonds. Such complexes resemble comb-shaped supramolecules that may self-assemble on a length scale of a few nanometres and generate mesophase materials.[2]
In this work we describe the preparation of individual cylindrical supramer - complexes that can exist independently of macroscopic phases. The supramers consist of polybases and wedge-shaped amphiphilic acids, which are able to self-assemble into cylindrical supramolecular structures.[3] The formation of such cylindrical structures should predominately be driven by the ionic interaction between the polymer segments and the low molecular weight acids, as well as the subsequent self-assembly of the wedge-shaped side groups around the macromolecular core and not by the entropic repulsion / excluded volume effects of the side chains as known with comb-like molecules bearing flexible linear side chains.[4] Hence, the supramers will mimic the architecture of a tobacco mosaic virus, in which a RNA chain is surrounded by self-assembled protein molecules.
The chemical structure of one of the complexes is depicted in Figure 2.1a. The synthesis of this compound was described elsewhere, both sodium and pyridinium salts of the wedge shaped acid (1-H) exhibited liquid crystalline phases.[3] The formation of complexes between 1-H poly(vinylpyridines)s (P2VP and P4VP) was monitored by means of FTIR, XPS as well as UV-VIS spectroscopy. Visualization of the single complex supramers on highly oriented pyrolythic graphite by scanning force microscopy was achieved at the degree of neutralization 25 mol% (Figure 2.1a). At higher content of 1-H incomplete complexation of sulfonic acid has been observed. It seems that 25 mol% substitution of P4VP by the bulky, wedge-shaped 1-H molecules is sufficient to form a densely packed 2-D pattern on HOPG. The UV-Vis spectroscopy showed a clear hypochromic effect upon increasing the degree of neutralization of P4VP (Figure 2.1b), indicating mutual interaction of the chromophores in the complex. Liquid crystalline order was observed in the complex with the degree of neutralization as low as 12.5 %. The dependence of the mesophase structure as well as the photochemical behaviour on the degree of neutralization will be discussed.
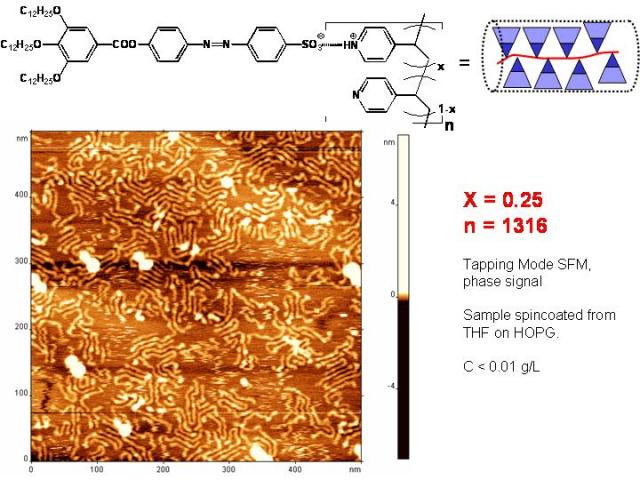
Figure 2.1a: Chemical structure of a complex formed from the wedge-shaped sulfonic acid molecule 1-H and P4VP and SFM image of the 1-H/P2VP complex with a degree of neutralization 25 mol%.
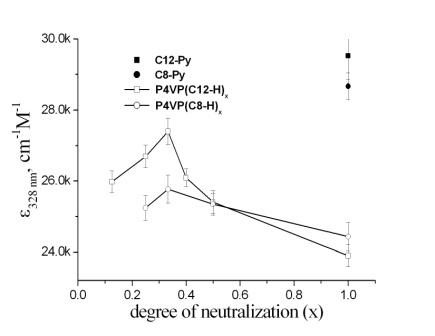
Figure 2.1b: Dependence of the molar extinction coefficient at 330 nm of the azo-chromophore on the degree of neutralization of P4VP
References
- K. Albrecht, A. Mourran, X. Zhu, T. Markkula, J. Groll, U. Beginn, W. H. de Jeu, M. Möller, “Thin Film Morphologies of Block Copolymers Complexed with Wedge-Shaped Liquid Crystalline Amphiphilic Molecules”, Macromolecules 41, 1728-1738 (2008).
- X. Zhu, U. Beginn, M. Moeller, R. I. Gearba, D. V. Anokhin, D. A. Ivanov, “Self-Organization of Polybases Neutralized with Mesogenic Wedge-Shaped Sulfonic Acid Molecules: An Approach toward Supramolecular Cylinders.”, J. Amer. Chem. Soc. 128, 16928 – 16937 (2006).
- K. Albrecht, A. Mourran, X. Zhu, R. I. Gearba, J. Groll, T. Markkula, U. Beginn, W. H. de Jeu, D. A. Ivanov, M. Moeller, “Supramolecular approach to hierarchical structures in thin films of block copolymers complexed with liquid crystalline amphiphilic molecules”, PMSE Preprints 95, 475 – 476 (2006).
- O. Weichold, X. Zhu, U. Beginn, V. Percec, M. Möller, “Polyelectrolyte-Dendron Complexes: A Route to Template Polymers”, Polymeric Materials: Science and Engineering 91, 202 (2004)

